Carbon dioxide CO2
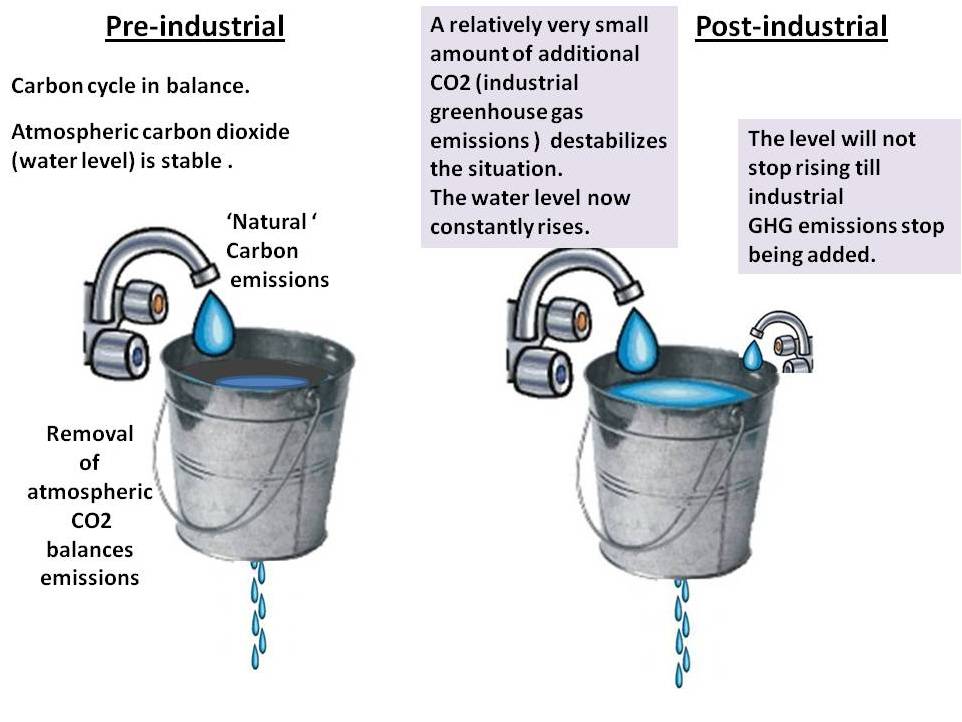
CO2 is so highly persistent and therefore cumulative in the atmosphere that so long as any extra 'unnatural' CO2 is being constantly added to the atmosphere it must build up/accumulate in the atmosphere.
The big thing about CO2 is that 'Carbon dioxide is (practically) for ever' (D. Archer 2008 Nature).
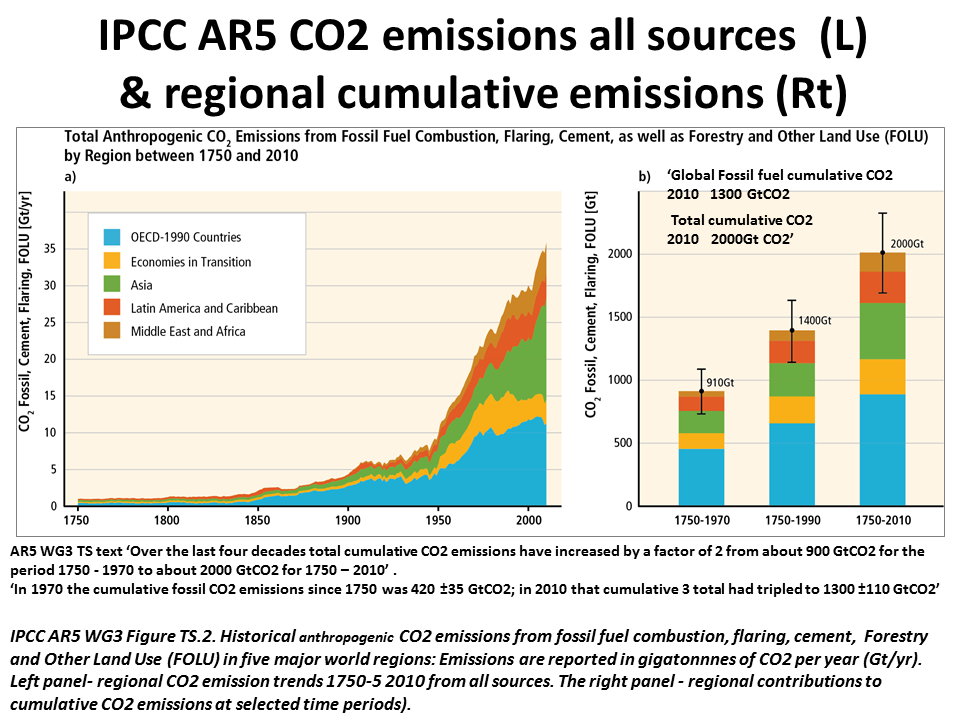
The answer is in what happens to all the CO2 being pumped into the atmosphere - constantly. Since 1750, 329 billion tons of carbon dioxide have been poured into the atmosphere from burning fossil fuels.
It does not just vanish into thin air. Like methane and nitrous oxide CO2 is called a' long lasting' GHG (i.e i the atmosphere). Most of it is still in the atmosphere and in the oceans where it is harmful to the living planet.
It does not just vanish into thin air. Like methane and nitrous oxide CO2 is called a' long lasting' GHG (i.e i the atmosphere). Most of it is still in the atmosphere and in the oceans where it is harmful to the living planet.
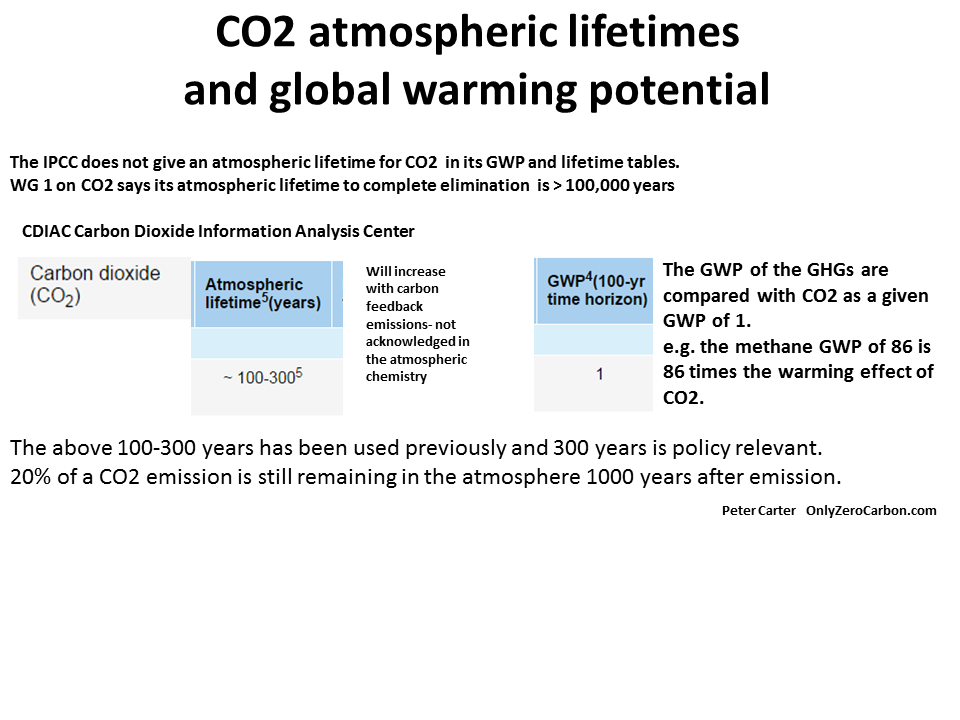
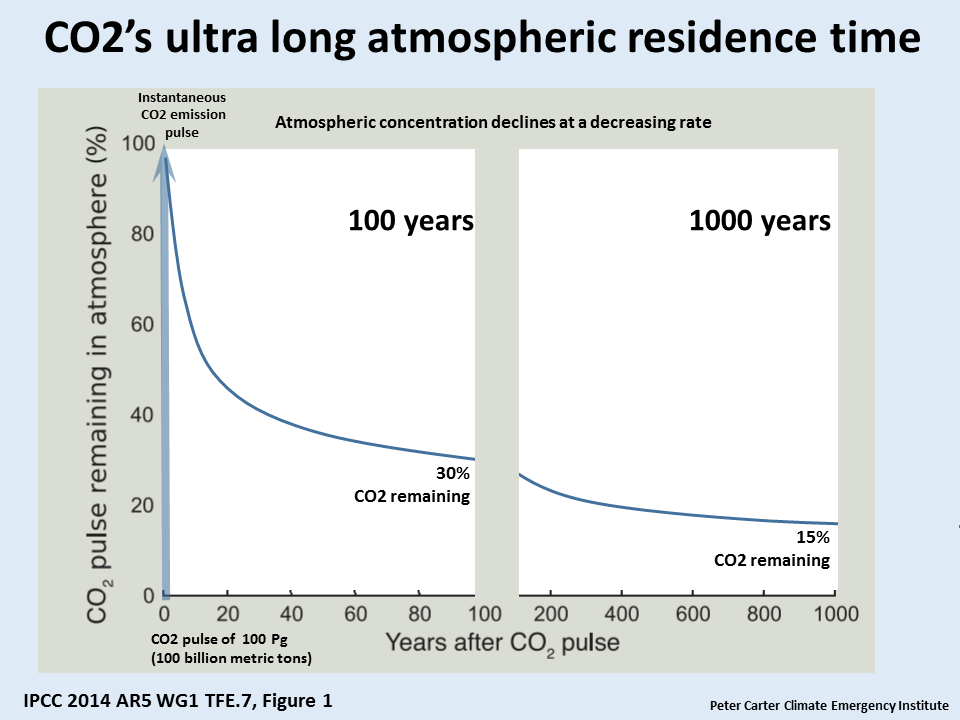
Why is this ? 20% or more of CO2 emissions last in the atmosphere for 1000 years.
The first graph shows the IPCC 2007 CO2 liftime from a computer model run that shows what happens to a single instantaneous pulse of CO2 emitted to the atmosphere. One thousand years after the CO2 is emitted to the atmosphere 20% is still there according to this model.
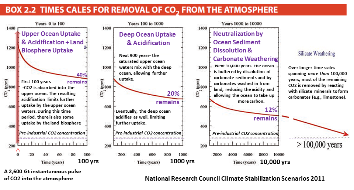
The next graph is from 2010 NRC Climate Stabilization Targets. Forty percent of the CO2 pulse disappears from the atmosphere in ten years- mainly being absorbed by the oceans. But after that, the removal of the CO2 pulse from the atmosphere is very slow. After one hundred years 40% of the CO2 is still in the atmosphere radiating heat energy.
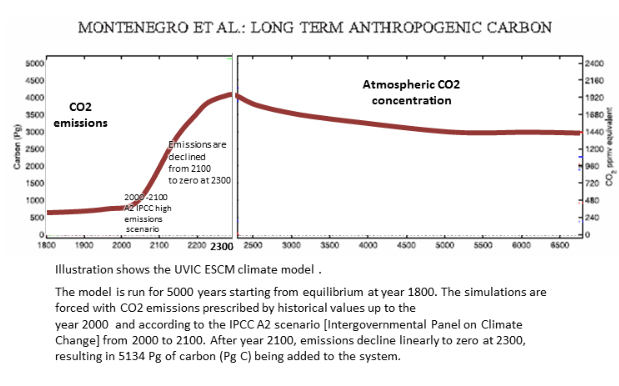
The last graph is from the Canada climate model including some carbon feedback and a realisitic reduction of emissions to zero. Here 75% of CO2 has a lifetime of 1800 years and 25% have lifetimes much longer than 5000 years.
The next graph is from 2010 NRC Climate Stabilization Targets. Forty percent of the CO2 pulse disappears from the atmosphere in ten years- mainly being absorbed by the oceans. But after that, the removal of the CO2 pulse from the atmosphere is very slow. After one hundred years 40% of the CO2 is still in the atmosphere radiating heat energy.
The last graph is from the Canada climate model including some carbon feedback and a realisitic reduction of emissions to zero. Here 75% of CO2 has a lifetime of 1800 years and 25% have lifetimes much longer than 5000 years.
The reason why so much of the carbon dioxide emissions stay in the atmosphere 'like for ever' is that our planet can only remove CO2 (really sink it) from the atmosphere in a permanent sense, which means removing from the carbon cycle at an ultra slow rate.
It takes thousands to hundreds of thousands of years for the planet to remove and sink carbon from the carbon cycle. Strictly speaking the atmospheric lifetime of CO2 emissions is 100,000 years.
It takes thousands to hundreds of thousands of years for the planet to remove and sink carbon from the carbon cycle. Strictly speaking the atmospheric lifetime of CO2 emissions is 100,000 years.
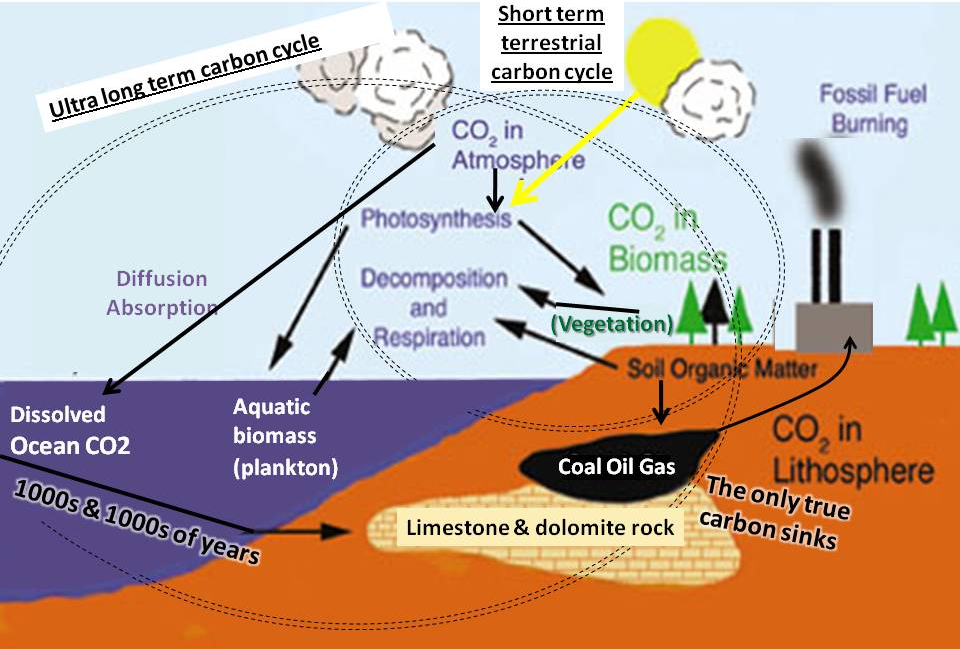
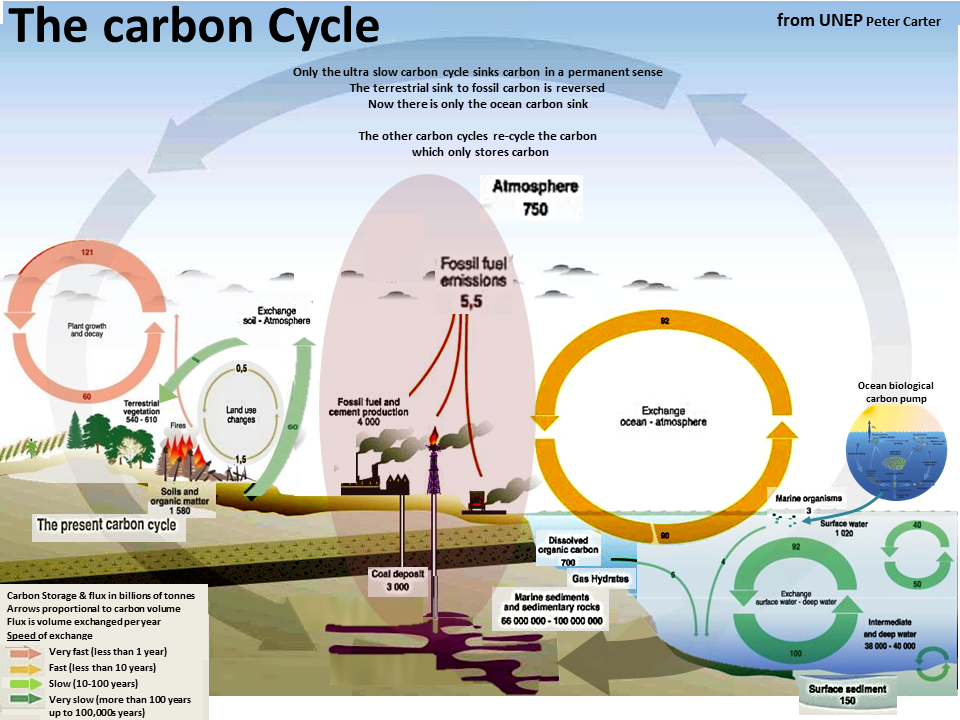
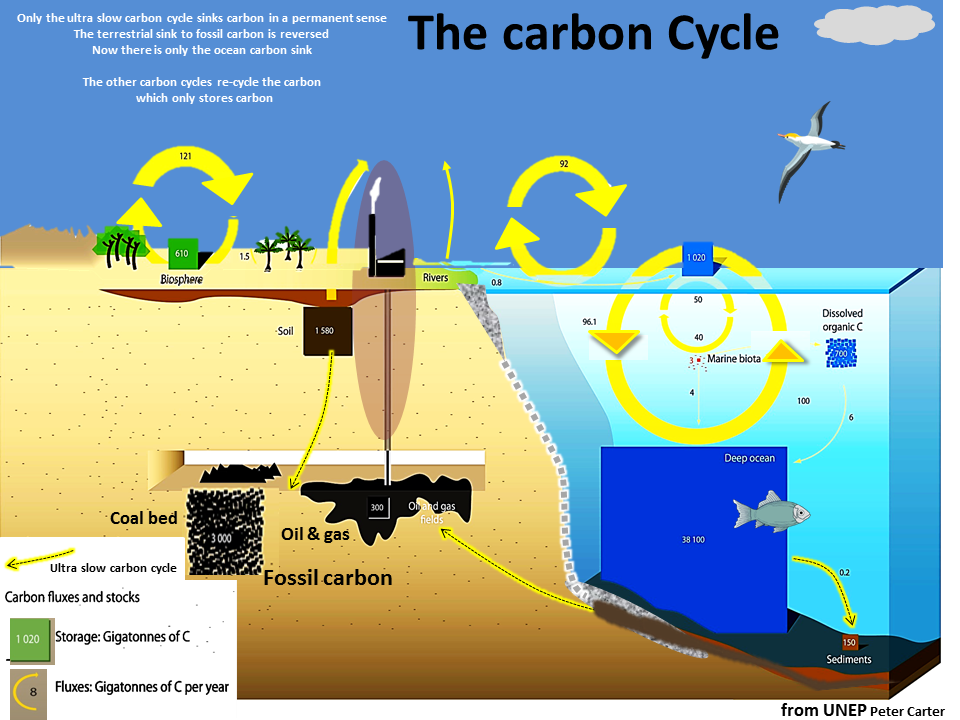
There are two ultra long-term/super slow carbon cycles by which the planet takes carbon out of atmospheric carbon dioxide.
One is land based and the other is ocean based.
They are both ultra slow processes happening over thousands to hundreds of thousands of years.
They both operate by dead organisms literally sinking and being compressed into liquid or sold matter.
One is land based and the other is ocean based.
They are both ultra slow processes happening over thousands to hundreds of thousands of years.
They both operate by dead organisms literally sinking and being compressed into liquid or sold matter.
The land sink forms what we call fossil fuels in the depths of the soil and rock.
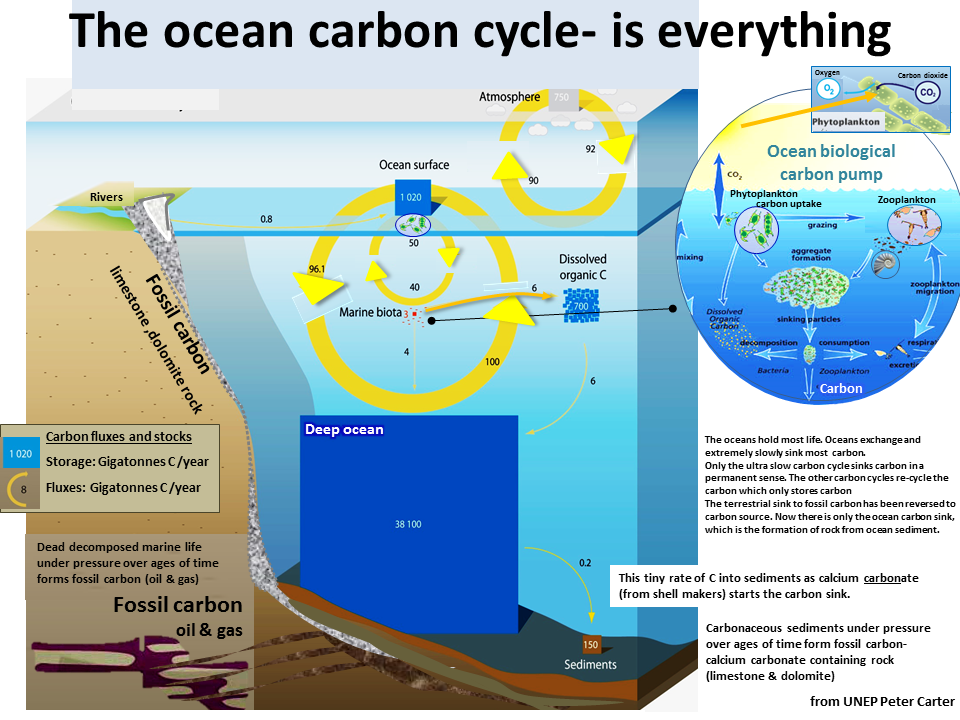
The same sort of thing happens deep below the ocean floor where dead marine organisms form limestone and dolomite rock which in the course of ages of time has formed rock on land.
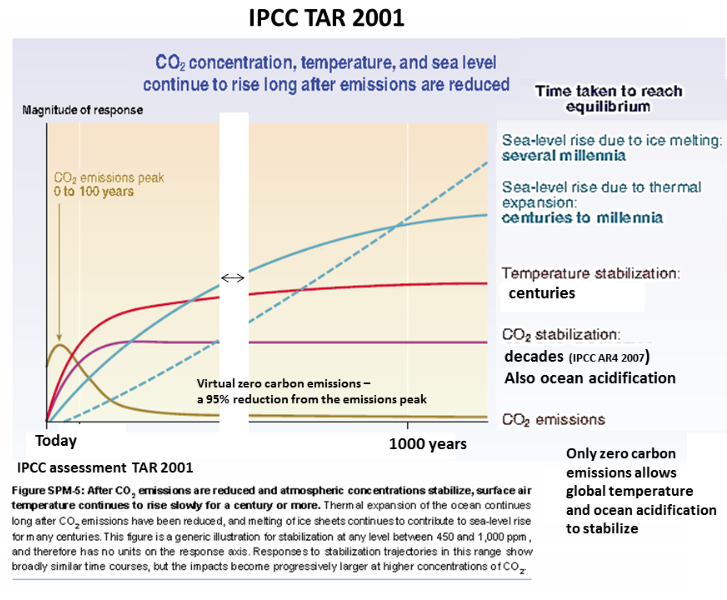
The IPCC graph is an illustration of climate change computer model runs. The computer model is run for the equivalent of 1000 years.
Starting at today on the graph global carbon dioxide emissions are rapidly reduced to virtual zero. The rate of the carbon dioxide emissions are constantly reduced till they reach virtual zero, which is about 90% on this graph.
Starting at today on the graph global carbon dioxide emissions are rapidly reduced to virtual zero. The rate of the carbon dioxide emissions are constantly reduced till they reach virtual zero, which is about 90% on this graph.
As emissions reduce the rate of increase of atmospheric CO2 slows to a plateau, but it never falls even with CO2 missions at a sustained virtual zero.
The rate of increase of global temperature lagging behind the slowing increase fate of atmospheric CO2. Even at 1000 years the temperature has not stopped increasing.
At 1000 years there is no sign of the rate of sea level rise slowing as land ice continues the melt more rapidly.
How then can we ever stop global warming and climate disruption?
First we have to stop emitting carbon dioxide. Everything else depends on doing that rapidly.
However even then there will be some increase in the carbon emissions, making the best that we can achieve virtual zero carbon emissions.
First we have to stop emitting carbon dioxide. Everything else depends on doing that rapidly.
However even then there will be some increase in the carbon emissions, making the best that we can achieve virtual zero carbon emissions.
To get to zero, or net zero, it is necessary to sink carbon over the very long term which means extract some carbon dioxide out of the air. This necessity of 'negative carbon emissions' or the development of 'artificial carbon sinks 'is another definite conclusion of the climate change scientific published research. A 2011 paper by J. Hansen and many other experts suggests that increasing the capacity or forests and agriculture to take up carbon could provide a carbon sink These measures certainly be tried and started right away. Biochar production from wood does have the potential to sink carbon for hundreds of years. The popular idea is to sink carbon by burning biomasss folr energy. It depends on the assumption that the CO2 from the burned biomass can be captured and stored away below ground (CCS) This is not a vlaid asumption and is not a good idea when we provide all out energy from zero carbon sources.
This graph published by the IPCC in its 2001 assessment shows all of the essential aspects of the climate change science that all results from the fact that umch of all carbon dioxide emitted to the atmosphere lasts there radiating heat for 1000 years.
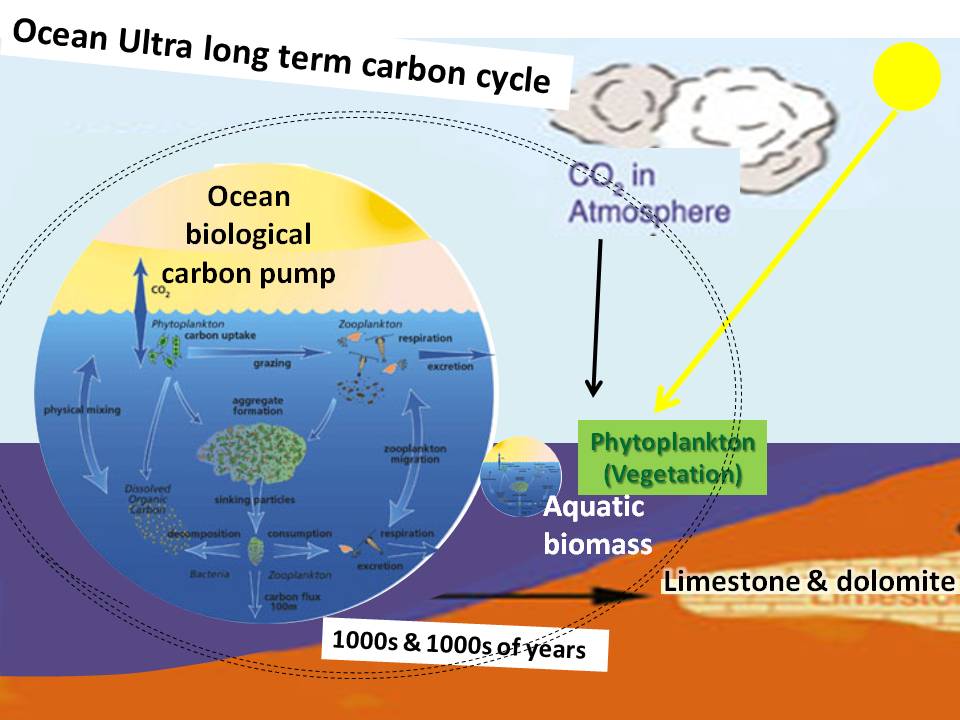
The ocean carbon sink process is initiated and depends on the plankton biological carbon pump.
Green phytoplankton take up about half of the total to carbon taken up by the planet's green plants.
The carbon captured by the phytoplankon becomes incorporated into shell bearing marine organisms that sinking to the ocean bottom are incorporated into the ocean sediments.
Green phytoplankton take up about half of the total to carbon taken up by the planet's green plants.
The carbon captured by the phytoplankon becomes incorporated into shell bearing marine organisms that sinking to the ocean bottom are incorporated into the ocean sediments.
only zero carbon only zero carbon only zero carbon only zero carbon only zero carbon only zero carbon only zero only zero
A CO2 molecule.
CO2 is a greenhouse gas because a CO2 molecule is an atomic dipole with an odd (3) number of atoms.
As such the CO2 molecule vibrates in the presence of infra-red emitting infra-heat energy itself.
CO2 is a greenhouse gas because a CO2 molecule is an atomic dipole with an odd (3) number of atoms.
As such the CO2 molecule vibrates in the presence of infra-red emitting infra-heat energy itself.
CO2 is an extremely long lasting atmosphericGHG.
For this reason, not only do CO2 emissions accumulate in the atmosphere, a very large proportion that will persist for 1000s of years is accumulating. In a practical sense that increasing proportion is there for ever.
A large proportion (over 30%) of all the hundreds of billions of tons of CO2 that has been emitted is in the oceans as carbonic acid and that will remain in the oceans for thousands of years.
CO2 is practically fore ever David Archer
The notion is pervasive in the climate science community and in the public at arge that the climate impacts of fossil fuel CO2 release will only persist for a few centuries. This conclusion has no basis in theory or models of the atmosphere/ocean carbon cycle, which we review here. The largest fraction of the CO2 recovery will take place on time scales of centuries, as CO2 invades the ocean, but a significant fraction of the fossil fuel CO2, ranging in published models in the literature from 20–60%, remains airborne for a thousand years or longer. Ultimate recovery takes place on time scales of hundreds of thousands of years, a geologic longevity typically associated in public perceptions with nuclear waste. The glacial/interglacial climate cycles demonstrate that ice sheets and sea level respond dramatically to millennial-timescale changes in climate forcing. There are also potential positive feedbacks in the carbon cycle, including methane hydrates in the ocean, and peat frozen in permafrost (D. Archer, 2-08, The millennial atmospheric lifetime
of anthropogenic CO2).
A study (Long term fate of anthropogenic carbon) including some of the carbon feedback finds the CO2 lifetime is even longer. About 75% of CO2 emissions have an average perturbation lifetime of 1800 years and 25% have lifetimes much longer than 5000 years.
CO2 is practically fore ever David Archer
The notion is pervasive in the climate science community and in the public at arge that the climate impacts of fossil fuel CO2 release will only persist for a few centuries. This conclusion has no basis in theory or models of the atmosphere/ocean carbon cycle, which we review here. The largest fraction of the CO2 recovery will take place on time scales of centuries, as CO2 invades the ocean, but a significant fraction of the fossil fuel CO2, ranging in published models in the literature from 20–60%, remains airborne for a thousand years or longer. Ultimate recovery takes place on time scales of hundreds of thousands of years, a geologic longevity typically associated in public perceptions with nuclear waste. The glacial/interglacial climate cycles demonstrate that ice sheets and sea level respond dramatically to millennial-timescale changes in climate forcing. There are also potential positive feedbacks in the carbon cycle, including methane hydrates in the ocean, and peat frozen in permafrost (D. Archer, 2-08, The millennial atmospheric lifetime
of anthropogenic CO2).
A study (Long term fate of anthropogenic carbon) including some of the carbon feedback finds the CO2 lifetime is even longer. About 75% of CO2 emissions have an average perturbation lifetime of 1800 years and 25% have lifetimes much longer than 5000 years.
Only zero carbon emissions can result in the stabilization of atmospheric CO2
Why is CO2 so persistent in the atmosphere?
The ultra long term slow carbon cycles that sink carbon
from the atmosphere
from the atmosphere
CARBON
o In the air - atmosphere
o On land - plants, soil, rocks
o In the air - atmosphere
o On land - plants, soil, rocks
o In the oceans
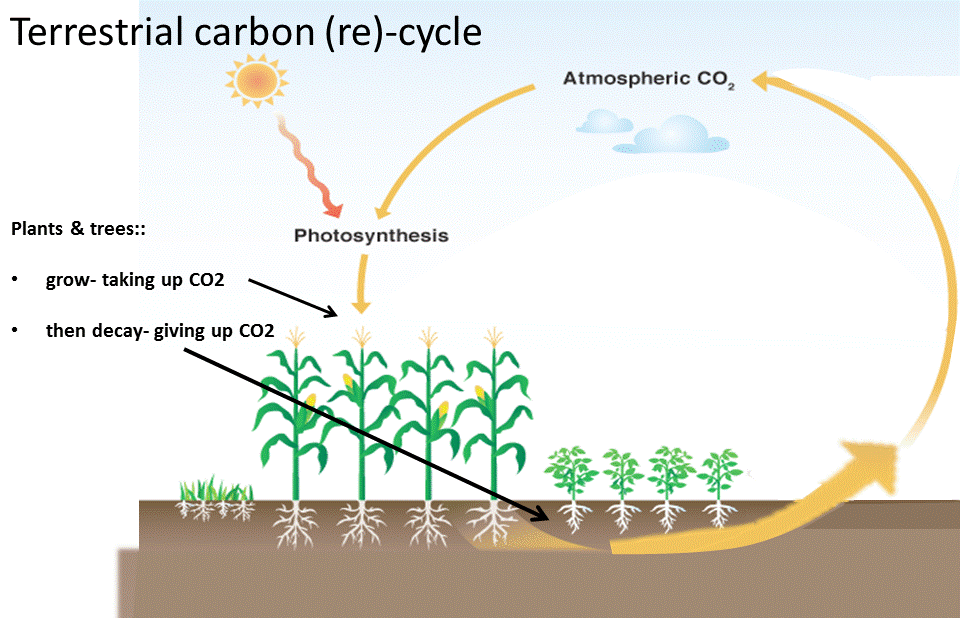
The carbon cycle on the land just continually re-cycles carbon.
There is no long term land carbon cycle anymore because we are burning fossil carbon.
Green plants absorb CO2 from the atmosphere to grow and when they die the CO2 is emitted back to the atmosphere. It is a short term carbon cycle that cannot help us in lowering the atmospheric CO2 concentration. Planting trees is essential for several environmental reasons but won't work for off setting carbon emissions.
If there was a massive global reforestation program atmospheric CO2 would be absorbed and could buy us a few decades to get to virtual zero and take carbon out of the air by other means.

The removal of human-emitted CO2 from the atmosphere by natural processes will take a few hundred thousand years (high confidence). Depending on the RCP scenario considered, about 15 to 40% of emitted CO2 will remain in the atmosphere longer than 1,000 years. This very long time required by sinks to remove anthropogenic CO2 makes climate change caused by elevated CO2 irreversible on human time scale. (IPCC AR5 WG1 Ch.6 Exec Summary
from IPCC AR5 WG1